Abnormal Bone Formation After Trauma Explained and Reversed in Mice
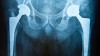
Ann Arbor, MI, USA – Hip replacements, severe burns, spinal cord injuries, blast injuries, traumatic brain injuries—these seemingly disparate traumas can each lead to a painful complication during the healing process called heterotopic ossification. Heterotopic ossification is abnormal bone formation within muscle and soft tissues, an unfortunately common phenomenon that typically occurs weeks after an injury or surgery. Patients with heterotopic ossification experience decreased range of motion, swelling and pain.